Biomanufacturing and Metabolomics
What is meant by biomanufacturing?
The Oxford Dictionary defines biomanufacturing as the process of using living systems, particularly microorganisms and cell cultures, to produce biological molecules and materials on a commercial scale; biomanufacturing uses living cells to produce medicine, proteins, antibodies, bioactive compounds, chemicals, textiles, and fuels.
Why is this an important field?
Sustainable manufacturing will replace various manufacturing that currently uses fossil and natural resources as raw materials or food sources as the world’s next generation of manufacturing and production. Biomanufacturing is important because it leverages biological systems to create novel products and sustainable alternatives, offering solutions across sectors like healthcare, materials, and energy. It also promotes economic growth and environmental benefits.
1. Addressing Global Challenges:
- Healthcare: Biomanufacturing is crucial in producing life-saving pharmaceuticals, vaccines, and therapies, including mRNA vaccines, cell and gene therapies, and antibodies.
- Sustainability: It enables the creation of sustainable materials and fuels from renewable resources, reducing reliance on fossil fuels and mitigating climate change.
- Economic Growth: Biomanufacturing fosters innovation, creates jobs, and strengthens domestic supply chains, contributing to economic stability.
- Environmental Conservation: It promotes eco-friendly practices, reduces industrial waste, and facilitates the development of green technologies.
2. Key Applications and Benefits:
- Biopharmaceuticals: Biomanufacturing is essential for producing complex biopharmaceuticals, including biologics and biosimilars, which are used to treat various diseases.
- Sustainable Materials: It offers opportunities to develop new materials with enhanced properties, such as biobased plastics and textiles, reducing dependence on petrochemicals.
- Renewable Energy: Biomanufacturing can produce biofuels and other renewable energy sources, contributing to a cleaner energy future.
- Food and Agriculture: It enables the development of new food products, crops, and agricultural inputs, improving food security and sustainability.
- Industrial Applications: Biomanufacturing can be used to produce a wide range of industrial chemicals, enzymes, and other products, offering alternatives to traditional chemical processes.
3. Advantages over Traditional Manufacturing:
- Sustainability: Biomanufacturing processes often require less energy and resources compared to traditional chemical manufacturing, reducing environmental impact.
- Efficiency: Biological systems can be engineered to produce specific products with high efficiency and precision.
- Flexibility: Biomanufacturing allows for the creation of a wide variety of products and materials, adapting to changing needs and demands.
- Reduced Costs: Biomanufacturing can lead to lower production costs, making products more accessible and affordable.
What are the challenges in biomanufacturing?
Biomanufacturing faces challenges including process complexity, scalability issues, regulatory hurdles, and ensuring product integrity and safety, all of which require careful optimization and innovation.
Here’s a more detailed breakdown of the challenges:
- Process Complexity and Variability: Biomanufacturing relies on living cells, which can exhibit high variability and complex interactions, making it difficult to control and optimize processes for consistent product quality.
- Scalability Challenges: Transitioning from laboratory-scale production to large-scale manufacturing can be difficult, requiring significant investments and adaptations to maintain product quality and yield.
- Regulatory Compliance: The biomanufacturing industry faces stringent regulations regarding product safety, manufacturing processes, and quality control, which can be complex and time-consuming to navigate.
- Product Integrity and Safety: Maintaining the integrity of biological products, such as proteins and antibodies, during manufacturing is crucial, as they are susceptible to degradation or contamination.
- Cost and Time: Developing and scaling up biomanufacturing processes can be expensive and time-consuming, requiring significant investments in research, development, and infrastructure.
- Ethical Considerations: The use of biotechnology raises ethical concerns, such as the use of genetically modified organisms, which require careful consideration and public discourse.
- Environmental Impact: The biomanufacturing industry can have a significant impact on the environment, requiring careful attention to waste management and resource utilization.
How can metabolomics be applied to the process of biomanufacturing?
Metabolomics can significantly impact biomanufacturing by enabling rapid identification of biomarkers, metabolic pathways, and process bottlenecks, ultimately accelerating strain engineering, optimizing bioprocesses, and improving product quality and yield.
1. Strain Engineering and Optimization:
- Identify Metabolic Bottlenecks: Metabolomics can reveal which metabolic pathways are limiting the production of desired compounds, allowing for targeted engineering efforts to improve efficiency.
- Optimize Growth Media: By analyzing the metabolic state of cells, metabolomics can help identify the optimal composition of growth media to maximize production.
- Develop Feeding Strategies: Metabolomics can help identify the optimal timing and type of nutrients to feed cells to maintain a healthy metabolic state and maximize product yield.
- Strain Selection: Metabolomics can help identify metabolic signatures that correlate with high-producing strains, aiding in the selection of the best candidates for biomanufacturing.
2. Bioprocess Monitoring and Control:
- Real-Time Monitoring: Metabolomics can provide a real-time snapshot of the metabolic state of cells during bioprocesses, enabling early detection of problems and adjustments to ensure optimal conditions.
- Biomarker Discovery: Metabolomics can identify new biomarkers that can be used to monitor the health and performance of cells and the quality of the final product.
- Process Optimization: By understanding the metabolic changes that occur during bioprocesses, metabolomics can help optimize various parameters, such as temperature, pH, and oxygen levels.
3. Product Quality and Safety:
- Identify Toxic Metabolites: Metabolomics can help identify toxic metabolites that may accumulate during bioprocesses, allowing for strategies to mitigate their effects and ensure product safety.
- Characterize Product Quality: Metabolomics can provide insights into the metabolic pathways involved in the production of specific products, helping to ensure that the final product meets quality standards.
- Develop Quality Control Methods: Metabolomics can be used to develop new quality control methods that can be used to assess the quality and safety of manufactured products.
4. Applications in Specific Areas:
- Biopharmaceutical Manufacturing: Metabolomics can be used to optimize the production of therapeutic proteins and other biopharmaceuticals.
- Industrial Biotechnology: Metabolomics can be used to develop new industrial processes for the production of chemicals and other products.
- Food Biotechnology: Metabolomics can be used to improve the quality and safety of food products including
cell-based meat products.
Plant Biotechnology: Metabolomics can be used to improve crop productivity and resilience to stress.
 Examples
Examples
Algae pathway
Triton Algae Innovations provides novel solutions for the world’s food and feed shortages and has taken the microalgae, Chlamydomonas reinhardtii, through the GRAS process. Chlamydomonas is safe, tastes great, and is rich in essential amino acids, omega-3 fatty acids, vitamin A, and other vital nutrients. To unleash Chlamydomonas’ potential, Triton developed scalable, environment-friendly manufacturing processes, demonstrated benefits to gut health, and pioneered biotechnology tools to produce nutritional compounds, such as colostrum proteins, for future foods.
In the development of Algae cultivars, HMT worked with Triton to better understand the biology of Algae and the effects of changing pH, UV light, and media on Algae biomass, production, and taste, including investigation of bitter-tasting development products.
To analyze food products, HMT utilized two platforms, our OMEGA Scan Advanced and LCOMEGA Advanced to monitor over 1,000 nutrients, metabolites, vitamins, and lipids from different cultivars.
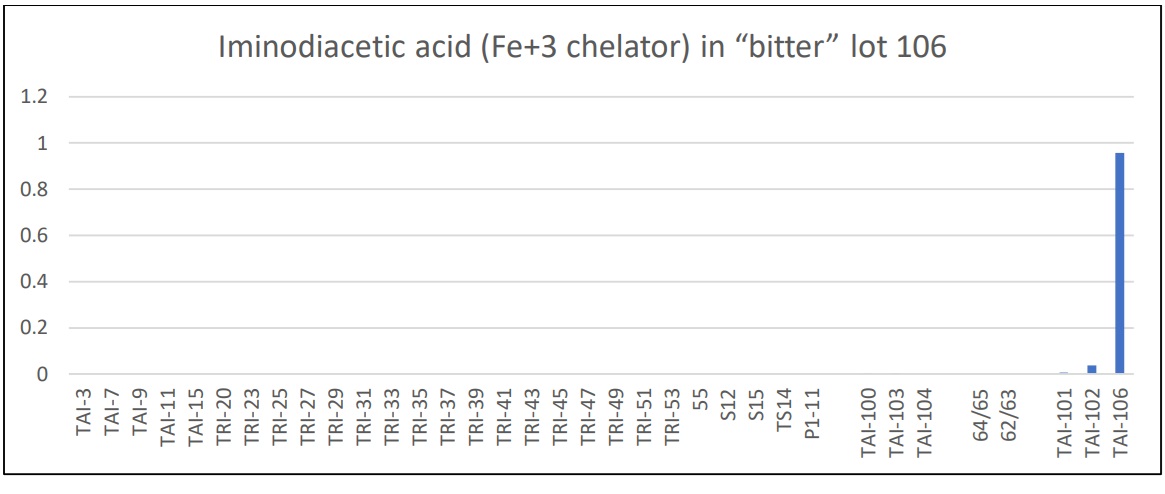
Lot 106 – Many chelators are organic acids and typically contribute to an acidic or sour taste. However, when complexed with a polyvalent metal (Mg, Mn, Zn) will provide a bitter taste. Compare the sour taste of citric acid to the bitter taste of Magnesium Citrate.
Lot 106 was found to have significant amounts of iminodiacetic acid, a known metal chelator, likely a candidate for the bitterness in lot 106.
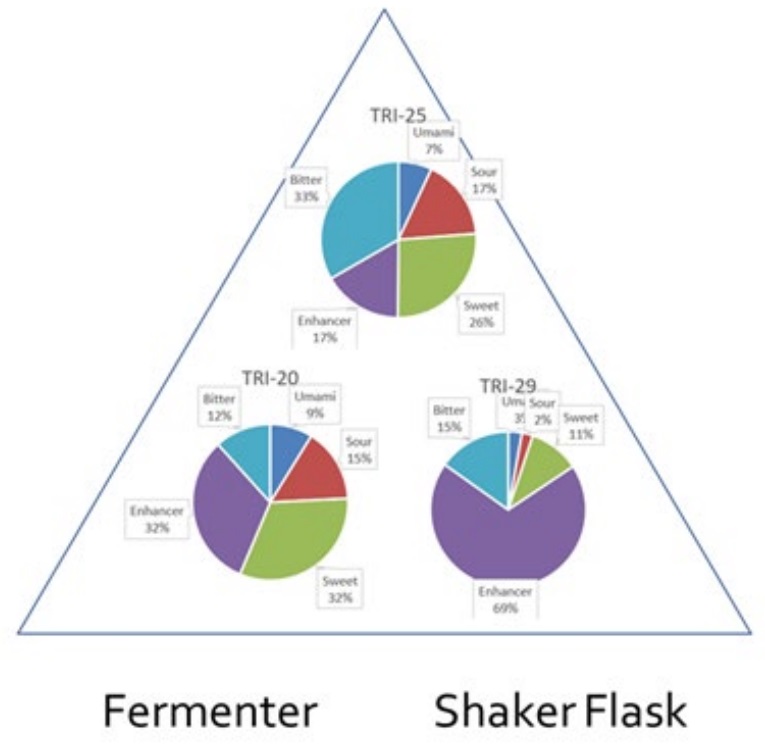
Triton was exploring moving from a shaker flask to a bioreactor or fermenter.
- The improved mixing and aeration possible in a bioreactor allow for higher cell densities. This can be equated to faster results or more material for a given volume of culture.
- The degree of measurement and control possible will always be greater. Standard sensors and peripheral equipment can make a bioreactor far more adaptable to user requirements. This increases the quantity of data available for analysis and feedback into process development.
- The design of a stirred tank bioreactor means they are often more compact than rockers and incubator shakers of equivalent working volume. As bioreactors normally produce higher yields, a smaller system may be all that is needed.
- The range of automated feeding strategies is greater. For example, using bags or shake flasks for continuous culture is rare.
Small pilot studies were then conducted, where metabolomics was applied to look at the overall composition of products including taste profiles:
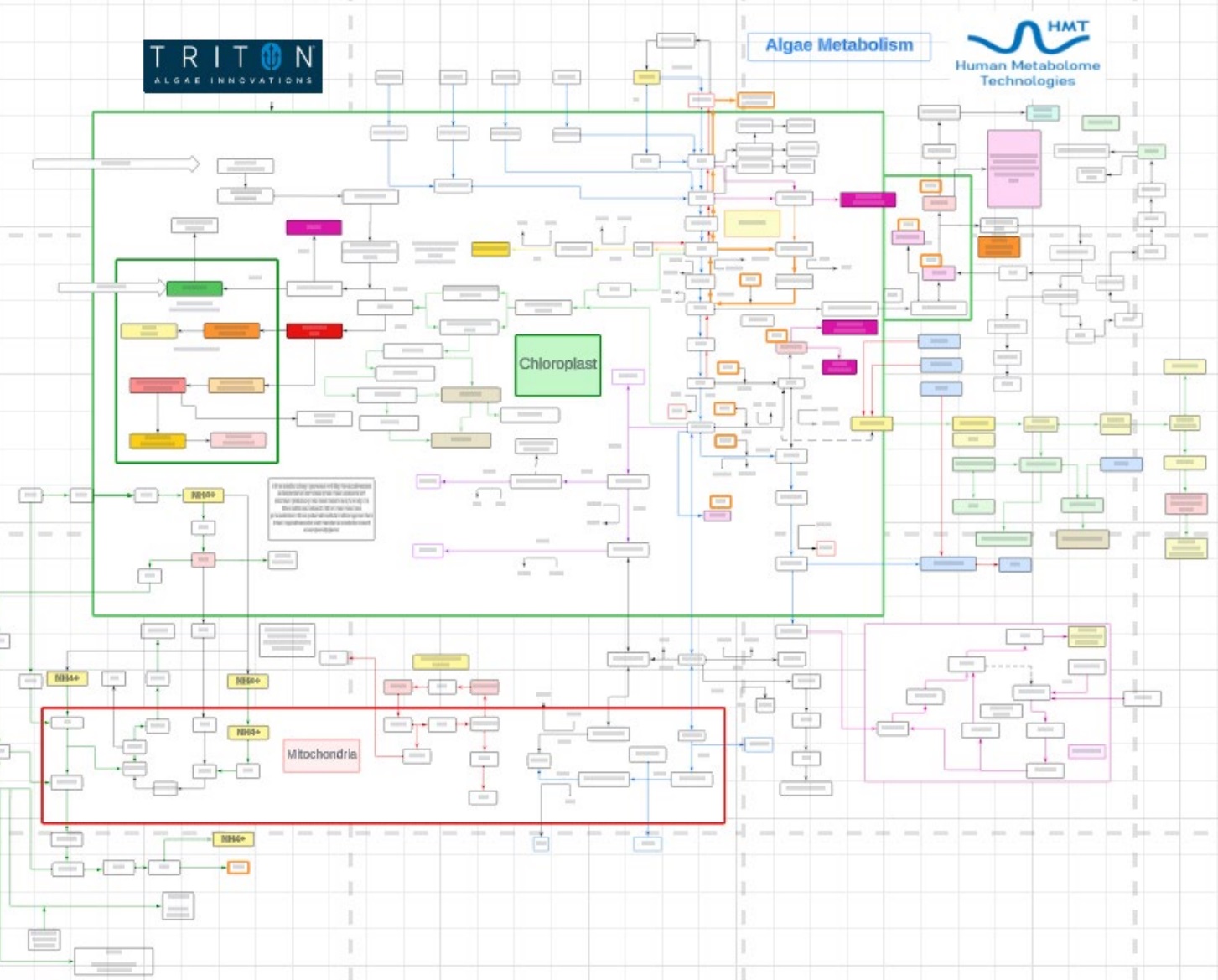
Lastly, different Algae products were fine-tuned using different combinations of UV light, pH, media composition, and time. Using comprehensive metabolomics and knowledge of biochemical pathways, a biochemical pathway map was developed to better understand the dynamics of each parameter as to different desired products such as carotenoids, biomass production, nutrient composition, and taste.
Terpene Production – Effects of metabolic engineering of the MVA pathway on
pleuromutilin production.
Saito et. al. elucidated candidate metabolic pathways to be modified for increased model terpene production by RNA-seq and metabolome analyses in A. oryzae and selected pathways such as ethanol fermentation, cytosolic acetyl-CoA production from citrate, and the mevalonate pathway. Changes in gene expression along specific pathways were measured against quantitative metabolomic profile (C-SCOPE) and product production.
*Saito, N., Katayama, T., Minami, A. et al. Versatile filamentous fungal host highly-producing heterologous natural products developed by genome editing-mediated engineering of multiple metabolic pathways. Commun Biol 7, 1263 (2024).
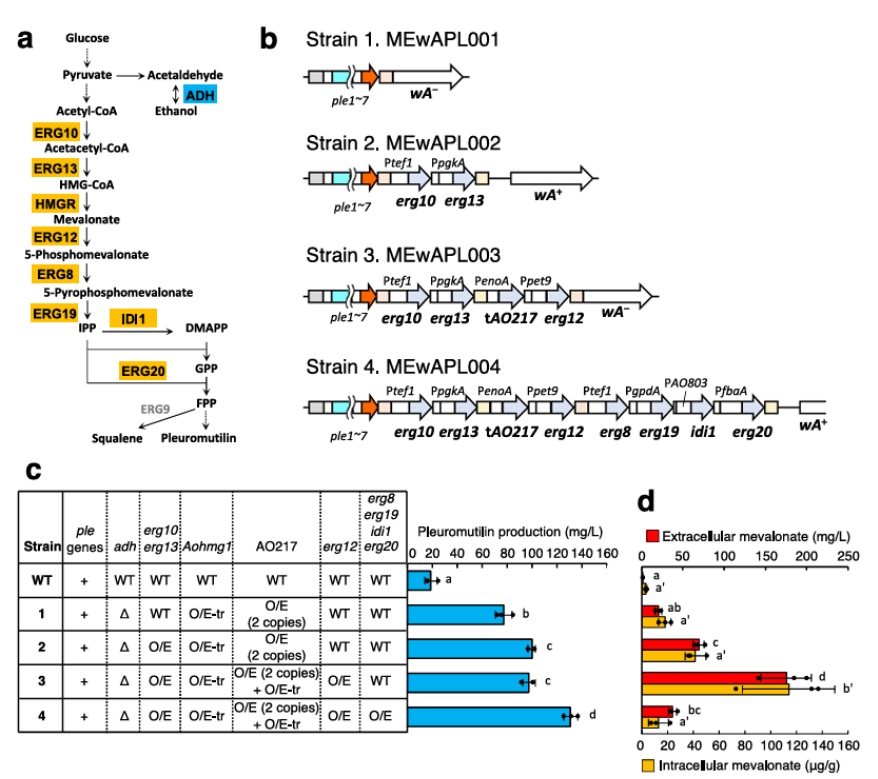
Effects of metabolic engineering of the MVA pathway on pleuromutilin production. (a) The ethanol fermentation and MVA pathways with the enzymes catalyze each reaction. The process with the protein indicated as blue box was blocked by gene deletion, and those with the proteins indicated as orange box were potentiated by gene overexpression. To activate HMGRs, their truncated variants were also overexpressed. B The cartoons indicate the wAloci of the indicated strains. Pleuromutilin production (c)and extracellular/intracellular mevalonate amount (d) in the indicated strains. The numbering of each strain corresponds to the strain numbering in the panel b. The strains were cultured in MPY liquid medium at 30°C for 5 days. “Δ” indicates deletion of adh, “O/E” indicates overexpression of the corresponding genes, and “O/E-tr” indicates overexpression of the truncated HMGR genes. Data are shown as means ± standard deviation (S.D.) with dot plot indicating each data of three independent experiments. Data are analyzed using Tukey’s(c) and Holm’s (d) multiple comparison test and means sharing the same letter are not significantly different (p > 0.05). The significance of intracellular mevalonate is indicated as a’ and b’ (d).
In this study, Saito et. al. elucidated the potential metabolic modifications in multiple metabolic pathways to stimulate the heterologous production of natural products using transcriptome and metabolome analyses in A. oryzae and demonstrated the effectiveness of these modifications on heterologous pleuromutilin production. Moreover, we engineered multiple metabolic pathways using CRISPR/Cas9 genome editing and generated host strain for heterologous terpene production containing a total of 13 metabolic modifications.
Conclusion
The world’s economy will be a bioeconomy, and it is HMT’s responsibility to help drive this transformation by supporting bioengineering in all ways possible.
